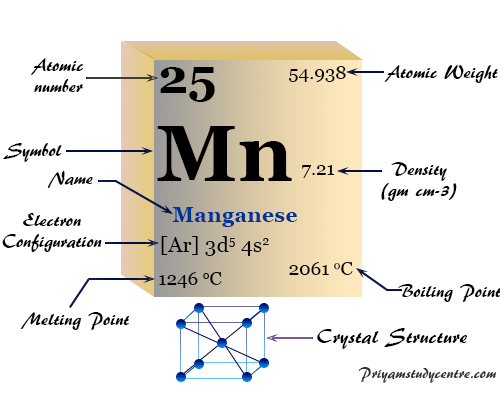
Mn element periodic table12/25/2023  Manganese minerals are widely distributed, with oxides, silicates, and carbonates being the most common. Recognized by Carl Wilhelm Scheele, Torbern Olof Bergman, and others as an element and isolated by Gahn in 1774 by reduction of the dioxide with carbon. 1995 - 2023.The mineral rhodochrosite is a manganese carbonate.ħ, 6, 5, 4, 3, 2, 1, −1, −2, −3 acidic, basic or amphoteric depending on the oxidation stateįrom the Latin word magnes, magnet, from magnetic properties of pyrolusite. Periodic Table of Elements - Manganese - Mn. 
If you need to cite this page, you can copy this text: This database focuses on the most common chemical compounds used in the home and industry. Molar mass calculations are explained and there is a JavaScript calculator to aid calculations. Molar Mass Calculations and Javascript Calculator.Introduces stoichiometry and explains the differences between molarity, molality and normality. Related ResourcesĪnswers many questions regarding the structure of atoms. Common Chemical Compounds of Manganese ReferencesĪ list of reference sources used to compile the data provided on our periodic table of elements can be found on the main periodic table page.Uses of Manganese: Used in steel, batteries, axles, rail switches, safes, plows and ceramics.Primary mining areas are South Africa, Russia, Gabon, Australia, Brazil. Annual world production is around 6,220,000 tons. Sources of Manganese: Most abundant ores are pyrolusite (MnO 2), psilomelane and rhodochrosite (MnCO 3).Name Origin: Latin: mangnes (magnet) Ital.70kg human: 12 mg Who / Where / When / How Note: this data represents naturally occuring levels of elements in the typical human, it DOES NOT represent recommended daily allowances. Target Organs: Respiratory system, central nervous system, blood, kidneys.Routes of Exposure: Inhalation Ingestion.Vapor Pressure = C Regulatory / Health.Flammablity Class: Metal: Combustible Solid.Enthalpy of Vaporization: 219.7 kJ/mole.Enthalpy of Atomization: 280.3 kJ/mole 25☌.Description: Silver-gray transition metal with a pinkish tinge.Conductivity Electrical: 0.00695 10 6/cm Ω.Coefficient of lineal thermal expansion/K -1: 22E -6.Valence Electron Potential (-eV): 220 Physical Properties of Manganese.Electronegativity: 1.55 (Pauling) 1.6 (Allrod Rochow).Electrochemical Equivalent: 0.29282g/amp-hr.Valence Electrons: 3d 5 4s 2 Electron Dot Model.Number of Neutrons (most common/stable nuclide): 30.Number of Electrons (with no charge): 25.Electrons per Energy Level: 2,8,13,2 Shell Model.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
